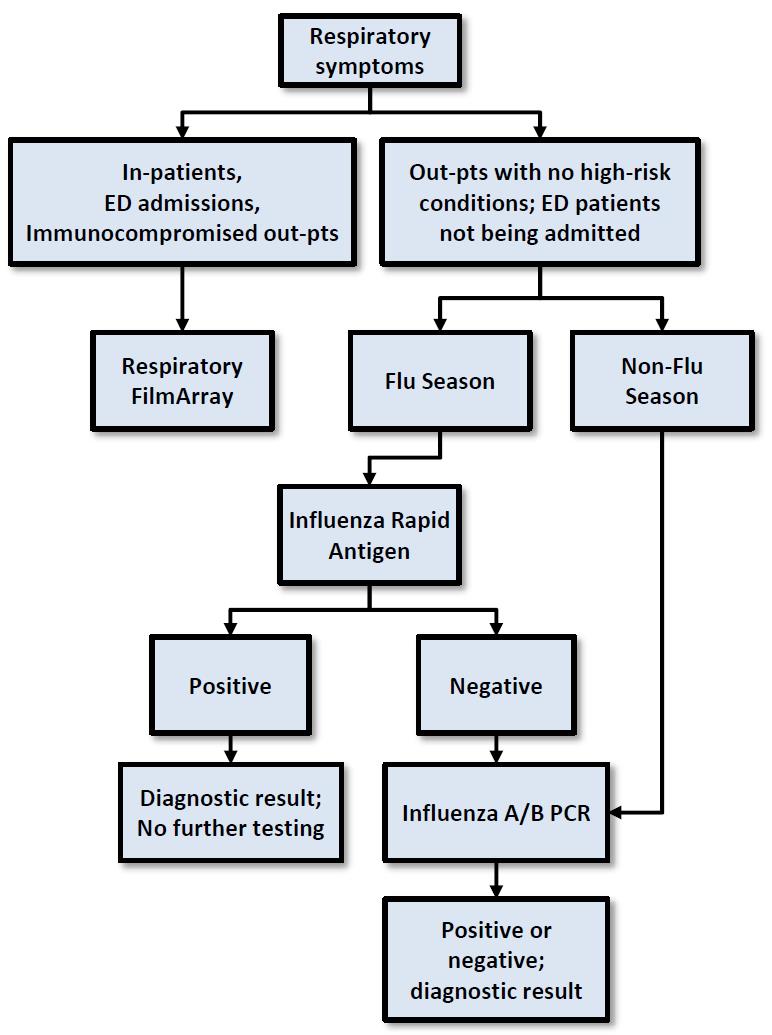
As we enter the winter months, there are often questions about the various respiratory pathogen tests available, including a multiplex molecular panel and influenza testing.
The Respiratory FilmArray® panel is a multiplex PCR assay that detects 20 different viral and bacterial pathogens (see table below). The rapid result provided helps to guide patient isolation precautions and proper antibiotic stewardship. As a relatively expensive test, this panel is most appropriately utilized for higher risk patient populations including inpatients, those being admitted by the Emergency Department, and immunocompromised outpatients (see figure below). This molecular test has been validated to test either nasopharyngeal (NP) swabs or bronchoalveolar lavage (BAL) fluid.
There is currently little literature regarding the relative performance of this assay when testing NP swabs vs. BAL fluid, however, to ensure optimal detection collect NP swabs for patients with primarily upper respiratory tract infections and BAL for patients with lower respiratory tract infections.
Please do not send both NP and BAL specimens as this is considered redundant testing and will not be performed on subsequent samples if collected within 7 days. Repeat testing within 7 days from either specimen type will only be considered based on acute changes in patient presentation since original testing (e.g. a NP swab was originally tested, but a patient develops a severe lower respiratory infection a few days later). Contact the Microbiology Lab (616.267.2640) in these situations.
| Respiratory FilmArray® panel targets | ||
| Viruses | Bacteria | |
| Adenovirus | Influenza A2 | Bordetella pertussis |
| Coronavirus1 | Influenza B | Chlamydophila pneumoniae |
| Human Metapneumovirus | Parainfluenza virus 1-4 | Mycoplasma pneumoniae |
| Human Rhinovirus/Enterovirus | Respiratory Syncytial Virus | |
1 Serotypes HKU1, NL63, 229E, and OC43.
2 Serotypes H1, H3, and H1-2009.
Influenza testing is comprised of a rapid antigen test and a molecular PCR test, with seasonal variation in testing required to optimize test performance.
The rapid antigen test can be used to quickly diagnose patients during times of high disease prevalence within the local community, but suffers from lower sensitivity (false negatives) and specificity (false positives) during warmer months.
The molecular PCR method provides a definitive result outside of “influenza season” and following negative antigen tests. Influenza testing should be ordered for outpatients with corresponding clinical symptoms. See previous article for more information regarding the utilization of influenza rapid antigen vs. molecular testing.
See the laboratory catalog for more ordering information: