Effective March 17, 2024, Corewell Health Advanced Technology Laboratory will update their CMV and EBV Quantitation testing.
The following information was updated in the Lab Catalog.
Effective Tuesday, February 20, 2024, the policy for home collected specimen drop offs for Outpatient Laboratories (“draw sites”) has been updated.
For all Corewell Health West Michigan locations: after registration, patients or their designated person will verify the specimen at the laboratory. Laboratory team members will verify the specimen is properly labeled and ensure they have everything needed to be able to test the specimen before the patient or their designated person leaves the building. This does not include specimens collected by a home health nurse or provider.
This will standardize our patient flow when arriving with a home collected specimen and will reduce the need for recollection due to missing information or improper collection.
When specimen collection orders are placed, please notify patients of the change to this process.
RELATED RESOURCES
Effective February 7, 2024, at 7:30 AM, Corewell Health West Laboratories will go live with a new reference range for the calculated Anion Gap (AG). The AG reference range will be adjusted to align with recent reference range studies.
The following will be the new normal reference ranges for AG:
Anion Gap: 5 – 14 mmol/L
The following information was updated in the Lab Catalog.
All changes are for Corewell Health Laboratories West.
As of January 26, 2024, for the following tests, the testing platform has changed from EliA Fluorescence Enzyme Immunoassay (FEIA) from Thermofisher to Multiplex Flow Immunoassay from Biorad. Please review the lab catalog for any changes to collection information or reference ranges.
• Anti-dsDNA Antibody
• Cyclic Citrullinated Peptide (CCP) Antibody
• Celiac Antibody Cascade
• Tissue Transglutaminase Antibody
• Gliadin Antibodies
Written by Yasel Fleitas Alvarez, Ph.D., Chemistry Clinical Advisor, Corewell Health Reference Laboratory West, Michigan Pathology Specialists.
This January we are celebrating the National Thyroid Awareness Month. In United States of America, it is estimated that approximately 20 million people have thyroid disease and most importantly, according to the American Thyroid Association (ATA) as many as 60% of people suffering from a thyroid disorder are not aware they have it.The thyroid is a butterfly shaped-gland located at the front of the neck that produces and release thyroid hormones (See Figure 1).
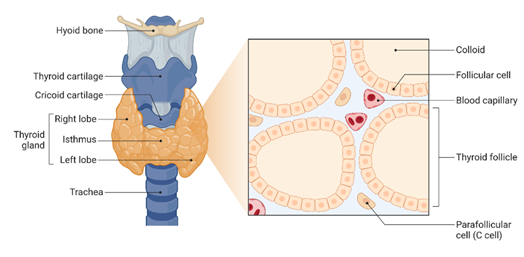
Figure 1. Thyroid Gland Anatomy and Histology
It regulates important physiological functions as:
-
- Breathing
- Heart rate
- Energy production
- Muscle strength
- Body temperature
- Weight
- Mood
Thyroid disease can present in two main forms:
- Hypothyroidism (under functioning thyroid)
- Hyperthyroidism (over functioning thyroid)
Confirmation or exclusion of thyroid disease requires a clinical examination combined with biochemical determination of thyroid hormones (TH) and thyrotropin (TSH)concentrations.
In this blog we discuss the best practices for ordering thyroid function tests for the initial screening of thyroid disease at Corewell Health.
Lab Specimen Label Printing Update for Corewell Health Epic Users
On 10/31/23, a change was made to lab specimen labels, which included a patient’s preferred name on the label, if it is documented in Epic. This highlighted a change in the names of label types seen by staff when reprinting specimen labels in Epic. Due to a mix of Intermec and Zebra printers across the entire Corewell Health system, defaults needed to be removed from Epic build to account for these differences. Users will now be required to manually choose the correct label type when reprinting labels. The Collection Activity behavior should remain the same.
As a reminder, referral testing, tests that are sent to another laboratory to perform, is dependent on the weather. Inclement weather can affect both the turnaround time of testing, and the testing capabilities of the organization. Our referral testing is dependent on outside transportation facilities (such as FedEx and UPS). When those organizations face challenges in weather, our testing capabilities must remain flexible.
If there is inclement weather in and around Memphis, TN (the main FedEx hub), there may be delays and cancellations of testing. We will try our best to communicate these changes as soon as possible via our blog: https://lab.spectrumhealth.org/
For any questions or concerns please “Contact Us” via the link above.
Effective January 17, 2024, UFH Anti-IIa testing will be performed at both Corewell Health Reference Laboratory West and Blodgett Immediate Response Laboratory in Grand Rapids.
