UPDATED 6/3/2020 – This test has been put on hold until further notice. Spectrum Health Laboratory will continue to forward orders for Noninvasive Prenatal Screening to Natera to perform the testing.
2020
Effective April 30, 2020, Spectrum Health Laboratories will no longer be performing the D-Dimer Semi-Quantitative assay. D-Dimer (quantitative) will be the only orderable for the D-Dimer assay. This D-Dimer will be reportable up to 35,000 ng/mL FEU. The cut-off value used in the diagnosis of venous thromboembolism (VTE), deep vein thrombosis (DVT), or pulmonary embolism (PE) remains at 500 ng/mL FEU. It is important to note inpatients can be (falsely) elevated in many underlying non-thrombotic conditions common in hospitalized patients.
Questions and inquiries regarding this change may be sent to the Coagulation Department using the “Contact Us” link above.
TEST INFORMATION
D-Dimer – [Epic Code: LAB313, Interface Code: 10123, CPT 85379]
Discontinued: D-Dimer Semiquantitative for DIC [Epic Code: LAB760, Interface Code: 10122, CPT: 85378]
Immunochemistry ANA (Antinuclear Antibodies) Testing Name Update
Effective April 22nd, 2020, the following ANA (Antinuclear Antibodies) Tests’ name will be updated. The change in naming convention comes in hopes of enhancing proper test utilization. This update includes only the name change with no changes in laboratory testing workflow.
| Old Test Name | New Test Name | Test Code | Clinical Utility |
| ANA (Antinuclear Antibodies) Screen, Double Reflex to ANA IFA Hep2 Titer and Pattern, and if positive to ANA antibodies | ANA screen | LAB1230491 | Preferred initial ANA screen for autoimmune connective tissue diseases. |
| ANA (Antinuclear Antibodies), Hep2 Substrate IFA with reflex to Titer and Pattern if positive | IFA ANA (Rheumatology)
|
LAB3540 | Preferred confirmatory ANA test for autoimmune connective tissue diseases. With ordering recommendations for rheumatology only. |
Test Update: Chromosome Analysis Breakage Study
Effective immediately, due to low test utilization and the high cost of maintaining proficiency, the Chromosome Analysis Breakage Study will no longer be offered by the Spectrum Health Cytogenetics Department.
If need arises for this test, please place an order for a Reference Miscellaneous (EPIC# LAB848) and enter “Test 140 DEB Breakage Study to Integrated Genetics” in the comments. The testing will be sent out through Spectrum Health Referrals Department to Integrated Genetics.
New posts will appear below this post. Reviewed 4/27/21, Last updated 4/27/21
PROVIDERS: Laboratory Testing COVID-19 Information
Effective March 4th, 2020, Thrombin Time will change from a sendout reference test performed by Mayo Clinic Laboratories to an in-house test performed by Spectrum Health Regional Laboratory. This test will be performed in the Coagulation Laboratory and will include a new reference range (please see link in Test Information below).
Thrombin Time is suitable for monitoring fibrinolysis therapy, screening for disorders of fibrin formation and in cases of suspected fibrinogen deficiency states. It is also useful for the differentiation between a heparin induced prolongation of the thrombin time and fibrin formation disorders.
For questions around this test, please contact the Coagulation Department at 616-267-2740.
TEST INFORMATION
Thrombin Time – Epic Code: #LAB11230581, Interface Code: #11230581, CPT: #85670
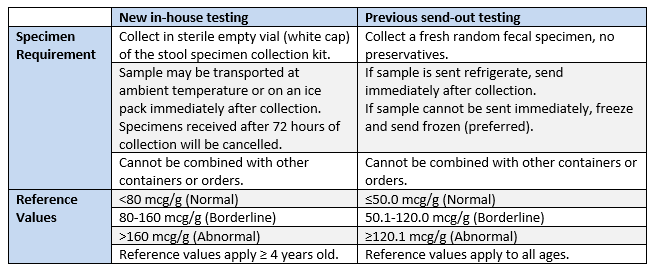
As of February 12, 2020 testing for fecal calprotectin will be brought in-house.
As result Calprotectin, Feces (LAB3290) will be replaced with Calprotectin, Feces (LAB1230580).
Please note the following differences between the new and previous test in terms of specimen requirements and reference ranges.
Questions may be directed to the Toxicology Laboratory using the “Contact Us” link above.